SCROLL DOWN
Foodborne Parasites: An Insidious Threat to Food Safety and Public Health
Often neglected in risk assessments, foodborne parasites deserve more scrutiny—and prevention
By Larry Keener, CFS, PA, PCQI
> FOODBORNE PARASITES
Image credit: Content Providers(s): CDC/DPDx - Melanie Moser
SCROLL DOWN
The list of foodborne parasites is long, and the morbidity and mortality they cause are very consequential. Yet, contemporary food safety literature on this subject is nearly mute. More often than not, the reporting on biological hazards in human food focuses on bacterial pathogens. Almost on a daily basis, we are inundated with reports of products recalled due to contamination with Listeria monocytogenes. The list of implicated foods is extensive: dairy products, fresh fruits, ice cream and frozen vegetables, ready-to-eat foods, and deli meats have all been the subject of extensive and costly recalls. The other bad actors in this biological hazard category include Escherichia coli O157:H7, Salmonella spp., Bacillus cereus, Campylobacter spp., Shigella, Staphylococcus, and even Clostridium botulinum. These disease-causing agents are so frequently reported in the contemporary literature and in news reports that even the lay public knows them by reputation. Recent data from the U.S. Centers for Disease Control and Prevention (CDC) confirm a public health burden for the United States of some 48 million cases of foodborne illness with 128,000 hospitalizations and upward of 3,000 deaths annually.1 The World Health Organization (WHO)’s global foodborne disease impact data for 2010 reported 600 million illnesses and 420,000 deaths. The WHO report states further that the preponderance of this public health burden was due to diarrheal disease agents, particularly norovirus and Campylobacter spp.2 But alas, there are insidious foodborne parasites that also tremendously increase the burden of disease on public health. The WHO also confirmed that the parasite Taenia solium is among the major causes of foodborne disease deaths globally.2
Cyclospora: A Case for Improved Identification Methods
Foodborne outbreaks of cyclosporiasis, for example, have been reported in the United States since the mid-1990s and have been linked to various types of imported fresh produce, including raspberries, basil, snow peas, lettuce, and cilantro. In 1996, there was a major foodborne disease outbreak occurring in raspberries imported from Guatemala. The outbreak was attributed to “small spherical bodies” observed via microscopy. “Small spherical bodies” is hardly scientific language for calling out the etiological agent in an ongoing disease outbreak, but in fact this is what was reported by CDC and U.S. Food and Drug Administration (FDA) investigators. It is a very common practice to use an organism’s primary morphological characteristics in parasitology, especially when dealing with parasites in food, water, or clinical specimens. The cartoon character-like face of Giardia intestinalis, under the microscope, is often used as a presumptive diagnosis for giardiasis, for example. The “small spherical bodies” in the 1996 raspberry outbreak, it turns out, were Cyclospora oocysts. Cyclospora oocysts, measuring 8 to 10 μm in diameter, are recognized as nonrefractile spheres (containing refractile globules) on light microscopic examinations of wet preparations of stool.3
Cyclosporiasis is caused by ingestion of the oocysts of Cyclospora cayetanensis, a parasite that was first recognized in the late 1970s. The clinical picture of cyclosporiasis is often indistinguishable from that of giardiasis, cryptosporidiosis, and isosporiasis. The main symptom is watery diarrhea that occurs in a relapsing, cyclical pattern, lasting for several days to weeks; it is often associated with weight loss and fatigue. In immunocompromised hosts, the duration and severity of illness may be increased.4 Between 2000 and 2017, there were 39 reported foodborne outbreaks of cyclosporiasis in the United States, involving a total case count of 1,730 individuals. In 17 of the outbreaks, food was identified as the vehicle of infection. According to FDA and CDC, identifying the particular food item or other ingredient that causes an outbreak of cyclosporiasis can be very challenging. CDC and other agencies are working to develop and validate molecular typing methods to distinguish among different strains of C. cayetanensis. In the future, such tools could help link cases of cyclosporiasis to each other and to particular types of produce, which could help public health officials investigate and prevent cases and outbreaks of Cyclospora infection.5
Epidemiologic studies have shown that parasite contamination of food and food ingredients may occur at several points along the supply chain, starting from the use of contaminated water for irrigation and contact with animal and human excrement during cultivation through contaminated harvesting equipment and including unhygienic practices of farmworkers or others handling the food material prior to consumption. Parasite transmission stages, like the oocysts of Cyclospora, tend to be robust and therefore likely to survive through the stages from harvesting to packaging to consumption. Foodborne parasitic diseases are often overlooked or neglected in various food safety control schemes, even though they are known to pose a severe threat to human health. Parasitic diseases in humans are notoriously difficult to detect, diagnose, and treat. This may account for this class of foodborne disease-causing agents being left out of the risk-assessment equation.
“…there are insidious foodborne parasites that also tremendously increase the burden of disease on public health.”
Common Food Sources of Parasites
Different types of parasitic diseases can be transmitted to humans by an assortment of food materials. Pork, freshwater crustaceans, sheep, fish, fresh fruit, and vegetables have all been implicated in parasitic disease outbreaks. Water and the environment have also been shown as viable, high-risk routes of human and farm animal infection. Food safety interventions, however, have proven effective in controlling or limiting the burden of foodborne parasitic diseases on public health. Thus, it is incumbent on food safety scientists to consider these parasites in the context of their risk assessment and food safety plans.
Trichinella: Meats
Porcine-transmitted parasites are well-known and very well documented. These include Taenia solium and Toxoplasma gondii. But perhaps of all the porkborne parasites, none is better known than Trichinella spp. Humans most often become infected with this parasite by consuming raw or undercooked meat containing the larvae of Trichinella. In the U.S., most often humans acquire the disease by eating raw or undercooked pork, wild boar, or bear meat. In other parts of the world, there are reports in the medical and public health literature of horse flesh as a source of transmission of this nematode to humans. Pork roast, smoked cured ham, sausages, and bacon are just some of the pork products frequently implicated in the transmission of this disease. Public health officials recommend heating pork or other Trichinella-harboring meat products to an internal temperature of 71 °C (160 °F) followed by maintaining that internal temperature (worst-case heating location) for a minimum of 2 minutes.6 The food safety concerns associated with foodborne parasites may further be exacerbated by the widespread usage of microwave ovens. It is interesting to observe that with the penetration of microwave ovens into Americans’ kitchens, there appears to be a corresponding increase in the number of cases of trichinosis (sometimes called trichinellosis). Between 1947 and 1967, CDC surveillance data show a precipitous overall decline in pig-mediated trichinosis in the U.S.7 The observed decline of this pernicious, parasitic disease in humans was due in large measure, according to public health officials, to changes in on-farm practices related to housing, feeding, and implementation of improved standards of care for swine prior to slaughter. Public health officials and veterinarians advised farmers against feeding the animals garbage and also of the need to prevent the exposure of pigs to rats—an important vector in the transmission of the disease. Concurrent with these on-farm developments, public health officials, home economics teachers, gym teachers, and librarians across the land were enrolled in an unprecedented campaign to educate the public on the perils of eating undercooked pork.
Between 1967 and 1979, CDC surveillance data showed large spikes in the reported cases of trichinosis in the U.S.7 The caseload levels observed during this interval were consistent with those observed in the 1950s. This period of increased disease activity corresponded with the increased uptake and use of microwave ovens by American consumers. According to J. Carlton Gallawa in A Brief History of the Microwave Oven, prior to 1967, only about 14 percent of U.S. households were using microwave technology.8 However, between 1975 and 1978, more than 60 percent of U.S. households owned microwave ovens. During this span, trichinosis cases went from fewer than 100 reported cases in 1967 to nearly 300 cases in 1975.9 This trend continued until about 1980, when the U.S. Department of Agriculture (USDA) issued guidelines for cooking pork in microwave ovens (internal temperature of greater than 170 °F).6 With the advent of these cooking guidelines, the trend line for the number of trichinosis cases fell off sharply so that they were again consistent with the observed caseloads prior to 1967 (Figure 1). These are not hard-and-fast data; rather, they are conclusions that have been drawn by various workers who have investigated the apparent anomalies in the downward trend of pigborne trichinosis in the U.S. These data are at least suggestive of a causal relationship between the microwave oven and the incidence of trichinosis.
USDA recommendations notwithstanding, it is generally recognized that microwave heating of foodstuffs is not uniform and consistent. The results of a study published in 1983 by W.J. Zimmerman in which he investigated the inactivation of Trichina in artificially infected pork roast showed that this lack of uniformity in heating resulted in the recovery of infective Trichina after cooking from 50 of 189 pork roasts.10 Viable Trichina was also recovered from a roast in which five temperature measurements from different areas of the roast exceeded 76.7 °C (170 °F). Zimmerman also reported the recovery of the infectious agent from pork roast that appeared to be well-done. Similar findings have been reported by Ribicich.11,12
In 2011, the USDA Food Safety and Inspection Service (USDA-FSIS) issued guidance that says eating pork cooked to an internal temperature of 63 °C (145 °F) with a 3-minute hold is an acceptable cooking standard for certain cuts of pork.13 They cite as justification for relaxing this public health guidance that the incidence of trichinosis has declined markedly in the last 50 years. In the late 1940s, there were about 400 cases each year; since 1982, there have been fewer than 50 a year, mostly traced to pork products eaten raw, or to wild game.14 The public health preventions and control measures that reduced rat populations on the farms and the prohibitions against feeding garbage to pigs are frequently cited as having contributed to a reduction or near elimination of the worm burden among commercially farmed animals. Most commercially farmed pigs in the U.S. are raised indoors. However—and a more contentious point of discussion about the application of the new guideline—what about animals raised outdoors where they are exposed to the parasite? Should the lower cooking standard apply equally to the meat derived from these high-risk animals? USDA-FSIS has not made a distinction. Some in the industry say that barnyard-raised hogs should not be risky when cooked using the new guideline. What about imported pork products? Is the risk profile for the imported meat equivalent to that of meat derived from domestic animals? We do know that trichinosis is a common zoonotic disease worldwide.
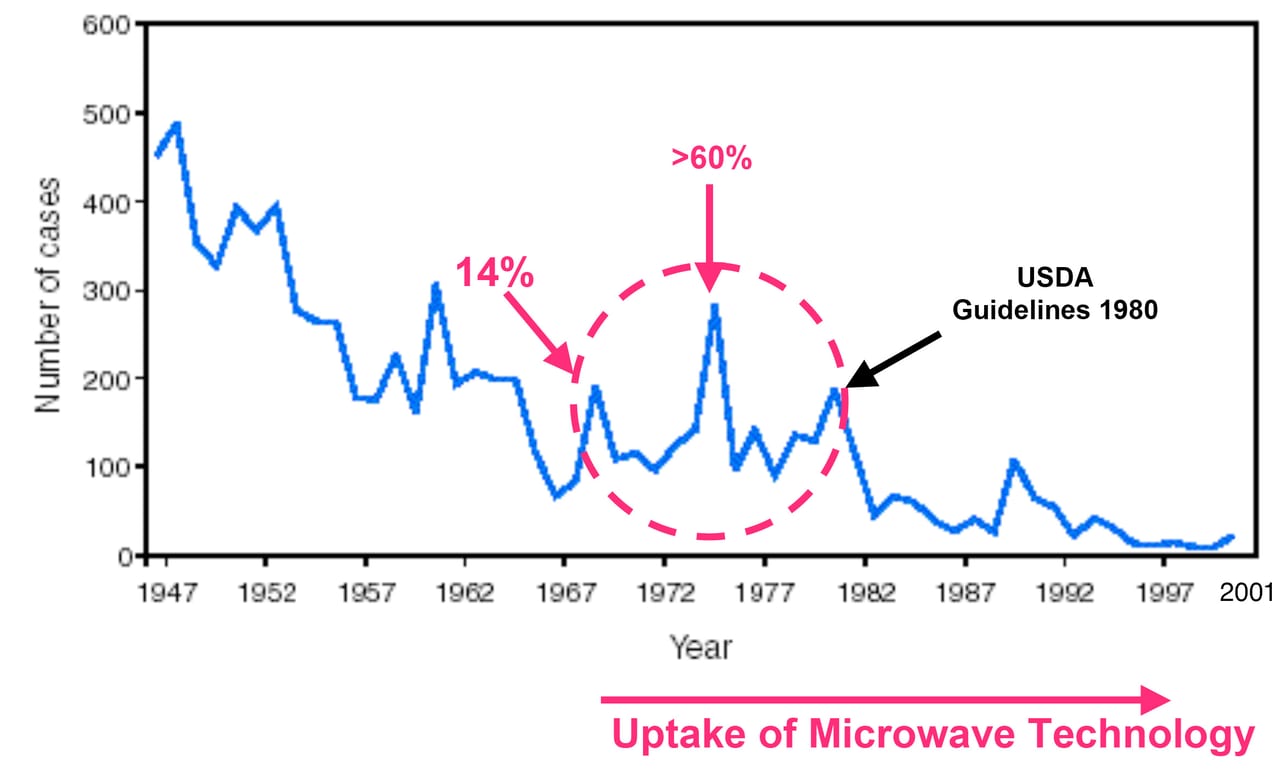
FIGURE 1. Number of Reported Cases of Trichinellosis by Year in the U.S., 1947–2001
Toxoplasma: Meats and Produce
Toxoplasma gondii (Isopora bigemina) is another important foodborne protozoan parasite that may be acquired by eating infected raw or poorly cooked meat, especially pig and sheep. There are also reports of T. gondii infections occurring as a result of eating poorly washed vegetables or vegetables that have been washed with T. gondii-contaminated water. The reproductive cycle of T. gondii occurs in Felidae and is particularly widespread in domestic cats.15 These carnivorous hosts become infected by eating meat contaminated with oocysts or other infective stages in the parasite’s life cycle. Rats and mice are reported as important vectors in the transmission of the disease in cats. Herbivorous animals acquire the infection by ingesting oocysts in food or water contaminated by the feces of infected cats. Oocysts of T. gondii are reportedly able to survive in the environment for up to a year. This parasite causes the disease toxoplasmosis.15
Toxoplasmosis is most common in warm, moist areas and is worldwide in distribution. It has been reported in man, pigs, sheep, cattle, dogs, cats, and other domestic animals. It is probably the most common infectious pathogenic animal parasite of humans, occurring in 30 to 50 percent of the human population. Serologic evidence indicates that 90 percent of the population of some areas is affected. Symptomology of the infection varies widely, but most cases of toxoplasmosis are asymptomatic.15 When symptoms are reported, they are similar to those of influenza. The main populations at risk of clinical disease are pregnant women and those with compromised immune systems. Infection during pregnancy has been reported to result in miscarriage or a stillborn infant.15 CDC reports that more than 40 million people in the United States may be infected with the Toxoplasma parasite.16
Preventing food sources from contact with feline feces or contaminated water is the best way to break the cycle of transmission. Cooking meat for 45 seconds at an internal temperature of 55 °C (131 °F) or for 6 seconds at an internal temperature of 63 °C (145 °F) or higher will kill the parasite. Freezing meat for 3 days at –20 °C (–4 °C ) is also effective at killing this organism.15 Caution should be exercised when washing fresh fruits and vegetables, especially those to be eaten raw, and preventive measures provided to make certain that the wash water has been maintained free from contamination by cat feces.15
Taenia: Pork
Taeniasis or tapeworm disease is caused by ingestion of the eggs of the pork tapeworm Taenia solium. As the name indicates, humans may acquire the disease from eating undercooked or raw pork. The reported incidence of infection in humans varies with the locality from less than 1 percent to about 8 percent, with a worldwide figure of 2–3 percent. The numbers worldwide may not be accurate due to confusion between T. solium and Taeniarhynchus saginatus (formerly T. saginata), the bovine tapeworm.15
I had my first encounter with T. solium during my time in service as a medical specialist during the Vietnam War. A young infantryman from the rural South presented at sick call with 2-foot-long worm that he had just recently passed in his stool. Having studied these creatures in parasitology doesn’t prepare one for an up-close-and-personal experience with this most unearthly looking creature. The infantryman, in this case, had been raised on a family-owned farm where it was the custom to keep barnyard swine as a source of food.
The pork tapeworm is common across most of Europe but rare in parts of Asia, the United States, and England. They are very common in Mexico. Humans are the only definitive hosts. In addition to pigs, camels, dogs, and monkeys are reported as intermediate hosts. The adult pork tapeworm lives in the small intestine and ranges in length from 2 to 7 m.15 The worms are passed in pig feces, and its eggs are ingested by humans and other animals. The muscle tissues of the pig may become heavily infected with cysts so that the meat is called “measly pork.”15 Again, humans usually acquire the adult tapeworm by eating uncooked or poorly cooked, infected pork.
Symptoms of adult tapeworm infection may be absent or mild. On occasion, there is diarrhea, weight loss, nervous symptoms, and even perforation of the intestinal tract. More serious symptoms are reported when the disease is reported as cysticercosis. Cysticercosis occurs when an immature but infective form of the parasite has penetrated the lumen of the small bowel and enters the bloodstream, where it is transported to other organs throughout the body.15 In this case, the extent and nature of the morbid processes are organ specific. Cysticercosis of the brain would obviously produce symptoms different from cysticercosis of the spleen, for example.
On-farm improvements, as mentioned earlier, have resulted in a dramatic reduction in the worm burden in commercially raised swine. Hogs raised as “free range” animals, however, remain vulnerable to this parasite.
“Epidemiologic studies have shown that parasite contamination of food and food ingredients may occur at several points along the supply chain...”
Anisakis: Marine Fish
Parasitic foodborne infections are by no means limited to terrestrial animals. Anisakis is an important foodborne disease-causing agent found in marine fishes. Anisakiasis, or herring worm disease, is a parasitic disease caused by nematodes (roundworms). The larvae of Anisakidae are commonly found in the mesentery and peritoneum of marine fishes. The disease, anisakiasis, is acquired by eating infected raw, pickled, or lightly salted fish such as herring, haddock, cod, mackerel, bonito, salmon, and pike.15 Undercooked squid have also been implicated as an etiologic agent of this disease. Symptoms are reported as similar to those of patients presenting with peptic ulcers or tumors, and they include nausea and vomiting with bloody diarrhea and fever.15 When people eat live herring worms, the parasite can be up to 2 cm in length; it invades the intestinal wall and causes symptoms, as reported above, that mimic those of foodborne illness, such as nausea, vomiting, and diarrhea. In most cases, the worm dies after a few days, and the symptoms disappear.15 This disease, also called anisakidosis, is rarely diagnosed because most people assume they merely suffered a bad case of foodborne illness, according to CDC. The global incidence of the disease is relatively low and limited in large measure to Japan and Korea, where eating raw fish and squid is a culinary habit. However, with the trend in many Western countries of eating sushi and related raw-fish dishes, it is reasonable to expect an increase in the incidence of anisakiasis worldwide.
A 2020 study led by scientists at the University of Washington and published in the journal Global Change Biology found “dramatic increases in the abundance of the parasitic nematodes Anisakis spp. and Pseudoterranova spp. in marine fishes and invertebrates.”17 These researchers reported an astonishing 283-fold increase in the abundance of these nematodes since the 1970s.17 Their work suggests implications for “the health of humans and marine mammals, which both can inadvertently eat the worms.” They looked specifically at the Anisakis spp. The University of Washington researchers don’t suggest a definitive cause for the massive increase of Anisakis worms over the past several decades, but they opine that climate change, more nutrients from fertilizers and runoff, and an increase in marine mammal populations over the same period could all be potential reasons.17
The worms can attach to the wall of the esophagus, stomach, or intestine. However, the Anisakis worm can’t reproduce or live for more than a few days in a human intestine. Humans are not its definitive host. But the worms can persist and reproduce in a number of marine mammals that serve as its intermediate host. The worms reproduce in the intestines of these animals and are released into the marine environment through excretion.15,18 Then the worms infect other creatures, including certain fish and squid that are used as human food. Another important implication of this study, for food safety scientists, is that now we know there is this massive, rising public health risk for those who consume raw or undercooked fish and squid. Is this new information reconciled in your food safety plan or risk assessment?
Phocanema: Marine Fish
A story of my personal encounter with nematodes in fish involves the so-called cod worm (Phocanema decipiens). Cod worms can infect various saltwater fish, especially cod, Pacific rockfish (also called Pacific red snapper), whiting, mackerel, haddock, herring, and salmon.15 I placed freshly washed cod fillets in a colander to drain and upon returning to cook them, 30 minutes later, I was greeted by a wriggling white worm emerging from its flesh. A model purchase specification proposes a maximum of three worms in 3.2 kg of fillets of whitefish in the United Kingdom, as judged by visual examination. An international standard, Codex Alimentarius, allows a maximum of five worms in 1 kg of fish of certain species.18 The moral of this story is, if you look, you will most likely find worms in your fish.
“Different types of parasitic diseases can be transmitted to humans by an assortment of food materials.”
Clonorchis and Paragonimus: Freshwater Fish and Crustaceans
Freshwater fish and crustaceans may also be reservoirs for parasites capable of causing disease in humans. Clonorchis sinensis and Paragonimus spp. are frequently cited in this group. C. sinensis is a fishborne parasite, whereas Paragonimus spp. is most often associated with freshwater shellfish.15,18
Clonorchis
C. (Opisthorchis) sinensis, also called the Chinese liver fluke, is the best known and best studied member of this family. The infection, named clonorchiasis, is caused by worms measuring 10–25 mm long and 3–5 mm wide; they are lanceolate, flat, and pinkish in color. The parasite’s prevalence in China is evident by the common name, but it is also found in Korea, Japan, and other Southeast Asian countries. Infection rates in local populations are reported to range between 15 and 70 percent. It is also important that in many areas, the infection rate among its many reservoir hosts may be as high as 100 percent. Reservoir hosts include dogs, cats, foxes, and badgers.15 Its life cycle is complex, involving several intermediate forms and at least two intermediate hosts, a snail and fish.15,18 It is not an uncommon practice among parasitologists to use primary morphological markers for the purposes of classification and identification. The eggs (27 × 16 µm) of C. sinensis feature a very distinctive hatch door (operculum) on one end of their lightbulb-like morphology.15 Cercaria, a juvenile, free-swimming form, emerges from the snail, penetrates the flesh of the fish, and becomes encysted, where they are then called metacercaria (0.1 mm in diameter). At least 80 species of fish in China, Korea, and Japan are classified as a secondary intermediate host of C. sinensis.15
Humans acquire clonorchiasis by eating raw, uncooked, pickled, or lightly salted fish.15 Infected fish contain encysted metacercaria; once consumed, they emerge in the small bowel of humans and migrate by way of the bile duct to the liver where they become adult flukes. The incubation period for the disease is about 3 weeks. Symptoms, when present, may include epigastric distress, diarrhea, vertigo, fluid accumulation in body tissues, and weight loss or wasting disease.15 Untreated infections are reported to persist for as long as 25 years or more, often resulting in an enlarged liver or a fatal type of cancer called cholangiocarcinoma.18 People can be treated for adult liver flukes using praziquantel, an anthelmintic drug. Anthelmintics are used for the treatment of worm infections in humans and other animals. A 1-day regimen of three 25 mg/kg doses given at 2-hour intervals is reportedly an effective treatment.19
The parasite can be killed by thorough cooking or freezing the fish. Cooking fish to temperatures of 70 °C (160 °F) or greater is lethal for encysted metacercaria. Freezing fish at –20 °C (–4 °F) for 7 days or at –35 °C (–31 °F) for 24 hours will inactivate the infective stages of the parasite. Freezing at temperatures above –20 °C will not kill the parasite. Inactivation by salting or pickling may or may not be effective, depending on the concentrations of these curing agents.18 According to FDA, nematode larvae can survive up to 28 days in 80 ° (salinometer) brine (21% salt by weight). Lime juice or vinegar at low concentrations, for example, is not effective at eliminating the parasite. Hot smoking when the internal temperature of the fish reaches 63 °C (145 °F) and is kept at that temperature for a minimum of 30 minutes is reported as an effective mitigation or control measure.
Paragonimus
Paragonimus spp. are frequently referred to in medical literature as lung flukes because of their propensity to attack this organ in susceptible mammalian populations, including cats, dogs, humans, and wild boar. P. westermani is one of the most important trematodes of man in some areas of the world, especially in the Far East.15 P. kellicotti is the only indigenous species in the U.S. The predominant Paragonimus species capable of causing human paragonimiasis by their geographic regions of distribution in the Americas are: P. caliensis (Central and South America), P. kellicotti (North America), and P. mexicanus (Central and South America).20
The life cycle of Paragonimus spp. is remarkably similar to that of C. sinensis, involving several intermediate and juvenile forms as well as at least two secondary intermediate hosts. The adult fluke measures 7–15 mm in length and resembles a small, reddish-brown coffee bean. The disease, paragonimiasis, is acquired in humans by eating improperly cooked or raw freshwater crab and crayfish containing its larvae.15,18 Once ingested, the parasite exits the small bowel, migrates to the peritoneum, traverses the diaphragm, and becomes established in the lungs. On occasion, the migrating larvae miss their intended destination and end up in the central nervous system and brain, resulting in severe complications for their victim. In the lung tissue, the mature fluke lays eggs (87 µm × 50 µm) that are both expectorated in the sputum and passed in the excreta of the infected individual. The operculated eggs have been reported in the sputum at concentrations high enough to tinge the infected person’s expectorate a rust color.15 The eggs, upon reaching freshwater, hatch, and the parasite’s miracidia, which are 0.08 × 0.04 mm in size, initiate a new life cycle.15
Paragonimus spp. are reported widely in tropical and subtropical regions; human cases have been seen only in limited areas of Asia, West Africa, and South and Central America where people have a custom of eating uncooked/undercooked freshwater crustaceans or wild boar meat. According to the WHO in 1995, approximately 20 million people were infected with lung flukes, and approximately 300 million people were at risk.21 The vast majority of patients as well as those at risk were in mainland China. Human infection with P. kellicotti has been reported in the U.S.22 P. kellicotti is endemic in U.S. freshwater river systems. Crayfish have been implicated in outbreaks of paragonimiasis in the U.S.22
Paragonimus spp. are destroyed by cooking techniques such as boiling. The recommended minimum inactivation time and temperature are 55 °C (131 °F) for 10 minutes. Freezing freshwater crab and crayfish at –20 °C (–4 °F) for a minimum of 48 hours will also inactivate this parasite. Pickling in salt brine, vinegar, or wine is not effective against this organism.18
“Fresh produce is frequently implicated in the transmission of foodborne parasitic diseases in humans.”
Conclusions: Give Parasites a Bigger Role in Threat Assessments
Fresh produce is frequently implicated in the transmission of foodborne parasitic diseases in humans. The infectious stages of both protozoan and helminth parasites have been reported on a wide variety of fresh produce and have been implicated as etiological agents in illness outbreaks reported worldwide. In such outbreaks, it appears that the fresh produce may have become contaminated with the parasites at any one of a number of points from the farm level to the consumer level, including the use of untreated water and application of animal or human feces to crops. Night soil—human excrement—when used to fertilize food crops, is a notorious source of parasitic disease in humans. Cestodes, nematodes, and trematodes have all been associated with diseases in humans, resulting from the use of night soil in agriculture. These helminth parasites include T. solium, Echinococcus spp., and Ascaris spp., for example. In 2015, the WHO Foodborne Disease Burden Epidemiology Reference Group identified T. solium as a leading cause of deaths from foodborne diseases.21
Protozoa may also be associated with diseases in humans that result from exposure to contaminated produce. In this scenario, the waterborne protozoa Entamoeba hystolitica and Cryptosporidium parvum are frequently cited in the outbreak literature. As previously discussed, Cyclospora has also emerged as an important disease-causing agent that has been associated with the consumption of contaminated fresh produce. A 2020 illness outbreak (ending in September 2020) reported by FDA, CDC, and 14 state health investigators confirmed more than 700 people with laboratory-confirmed Cyclospora infections. This outbreak attributed to bagged salad mix resulted in about 5 percent of the infected individuals being hospitalized. There were no deaths assigned to this outbreak. Another notable outbreak of cyclosporiasis occurring in 2013 and 2014 was linked to cilantro and salad mixes and affected more than 600 people. These incidents confirm that helminths and protozoa are important foodborne disease-causing agents in humans. Parasitic foodborne diseases are not limited to animal flesh, fish, and seafood. There is an abundance of data to confirm their transmission by way of fresh fruit and vegetables.
Risk assessment, with the passage of the Food Safety Modernization Act, has become the fulcrum of food safety assurance. All potential threats to public health that may associated with the consumption of human food must be identified, quantified, and mitigated when food safety is the objective. Primarily because of the public health burden attributable to foodborne diseases where bacteria are the etiological agents, most food safety plans have a bias toward bacterial agents, such as Salmonella, Listeria, and E. coli. This emphasis on bacterial pathogens appears to be justified based on the available outbreak and incidence data. However, in this category of biological foodborne agents are the foodborne parasites. Worms and protozoa are important in the transmission of foodborne disease in humans. Foodborne parasites affect the health of millions of people every year, infecting muscle tissues and organs, causing epilepsy, anaphylactic shock, amoebic dysentery, and other problems. Many can live in our bodies for decades. Despite their huge social costs and global impacts, information is generally lacking regarding just where these parasites come from, how they live in the human body, and—most importantly—how they make us sick. This information is critical for conducting a comprehensive and reliable risk assessment. In 2014, the WHO and the Food and Agriculture Agency of the United Nations (FAO) published a multicriteria-based ranking of risk for the management of foodborne parasites.23 The WHO/FAO risk ranking profile for the top 10 foodborne parasites and food sources is listed below:
- Taenia solium (pork tapeworm): In pork
- Echinococcus granulosus (hydatid worm or dog tapeworm): In fresh produce
- Echinococcus multilocularis (tapeworm): In fresh produce
- Toxoplasma gondii (protozoa): In meat from small ruminants, pork, beef, game meat (red meat and organs)
- Cryptosporidium spp.(protozoa): In fresh produce, fruit juice, milk
- Entamoeba histolytica (protozoa): In fresh produce
- Trichinella spiralis (pork roundworm): In pork
- Opisthorchiidae (family of flatworms): In freshwater fish
- Ascaris spp. (small intestinal roundworms): In fresh produce
- Trypanosoma cruzi (protozoa): In fruit juices
The scope and magnitude of several recent outbreaks associated with these pathogens infer that they are as consequential as the bacteria in terms of adverse public health outcomes and in preserving the integrity of the food supply. During the 1990s, it was customary, when attending conferences on foodborne disease, to have lectures on risk assessments and discussions about “emerging pathogens.” Based on this review of foodborne parasitic disease in humans, we suggest that a more rigorous and comprehensive risk assessment is warranted and that many types of food are increasingly vulnerable to this threat. The data suggest that attention must be given to those helminths and protozoa that are increasingly associated with a wide array of foods as multiple supply chains are implicated in their transmission to humans. There appear to be a number of factors involved in causing the emergence of foodborne parasites as a threat to public health. These include changes in culinary habits and more cosmopolitan food consumption patterns, population increases, declines in safe water sources, and projection of food supply chains into uncharted areas with no or underdeveloped food safety support systems. Further, as is suggested by current research, climate change may also be an underlying and contributing factor.
When the concept of Hazard Analysis and Critical Control Points (HACCP) was introduced to the food processing industry as a process control strategy, the Hazard Analysis step was intended to identify and quantify the magnitude of the threats to food safety. A quantitative or qualitative understanding of foodborne risk is the first requirement of an HACCP-based food safety control strategy. The second and perhaps the most daunting aspect of HACCP is the defining of the control measures that can reduce the foodborne parasitic threat to safe and acceptable levels. What are the current control measures and what are the current food safety objectives for foodborne-disease-causing parasites in human food? I will address those questions in next month’s issue.
References
- https://www.cdc.gov/foodsafety/foodborne-germs.html.
- https://journals.plos.org/plosmedicine/article?id=10.1371/journal.pmed.1001923.
- https://wwwnc.cdc.gov/eid/article/5/6/99-0604_article.
- https://www.cdc.gov/parasites/cyclosporiasis/outbreaks/foodborneoutbreaks.html.
- https://ask.usda.gov/s/article/To-what-temperature-should-I-cook-pork#:~:text=Cook all raw pork steaks,minutes before carving or consuming.
- CDC. 1986. “Trichinosis Surveillance, 1984.” Morb Mortal Wkly Rep 35: 11SS–15SS.
- www.smecc.org/microwave_oven.htm.
- Schantz, P.M. 1983. “Trichinosis in the United States–1947–1981.” Food Technol 37: 83–86.
- Zimmermann. W.J. 1983. “Evaluation of Microwave Cooking Procedures and Ovens for Devitalizing Trichinella in Pork Roasts.” J Food Sci 48(3): 856–860.
- Ribicich, M., et al. 2004. “Efficacy of Microwave Cooking to Inactivate Trichinella spiralis Larvae in Pork.” Veterinaria Argentina (21).
- https://pubmed.ncbi.nlm.nih.gov/11484370/.
- https://www.foodsafetynews.com/2011/05/usdas-new-recommended-pork-cooking-temp-145f/.
- https://www.washingtonpost.com/archive/lifestyle/food/1995/02/01/cooking-pork-what-about-the-old-rules/259cd15e-b33c-4380-ac85-5772881c3485/.
- Nobel, E.R. and G.A. Nobel, Parasitology –The Biology of Animal Parasites, 4th ed. (Philadelphia, Lea & Febiger, 1976).
- https://www.cdc.gov/parasites/toxoplasmosis/gen_info/faqs.html.
- https://doi.org/10.1111/gcb.15048.
- http://www.fao.org/3/cb1910en/CB1910EN.pdf.
- http://www.fao.org/3/x5951e/x5951e01.htm.
- https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/praziquantel.
- https://www.who.int/publications/i/item/WHO-UCN-NTD-VVE-2021.6.
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3719489/.
- http://www.fao.org/news/story/en/item/237323/icode/.
Larry Keener, CFS, PA, PCQI, is president and CEO of International Product Safety Consultants. He is also a member of the editorial advisory board of Food Safety Magazine.
